pan-Canadian Pharmaceutical Alliance (pCPA): August 2019 Trends and Insights
The pan-Canadian Pharmaceutical Alliance (pCPA) has issued the August 31, 2019 status of brand negotiations. Highlights since the last update include:
- 5 products completed CADTH review, for a total of 13 files under consideration;
- 8 products initiated pCPA negotiations, for a total of 47 active negotiations;
- 7 negotiations completed for a total of 270 completed negotiations;
- 1 negotiation closed, for a total of 44 closed negotiations; and
- 1 file closed without negotiations, for a total of 68 declined negotiations.
Completed
| Brand Name | Generic Name | Manufacturer | Indication | Initiation | Duration* |
|---|---|---|---|---|---|
| Imfinzi | durvalumab | AstraZeneca | Non-small cell lung cancer | Jul 2019 | 31 days |
| Cystadrops | Cysteamine | Recordati Rare Diseases | corneal cystine crystal deposits | Jun 2019 | 61 days |
| Tafinlar/Mekinist | Dabrafenib & Trametinib | Novartis | Melanoma | Jun 2019 | 61 days |
| Lonsurf | Trifluridine & Tipiracil | Taiho Pharma | Metastatic colorectal cancer | May 2019 | 92 days |
| Unituxin | Dinutuximab | United Therapeutics | In combination with GM-CSF, IL-2 and Retinoic acid (RA) for the treatment of pediatric patients with high-risk neuroblastoma | May 2019 | 92 days |
| Zirabev | Bevacizumab | Pfizer | Metastatic Colorectal Cancer | Mar 2019 | 153 days |
| Fibristal | ulipristal acetate | Allergan | Intermittent treatment of moderate to severe signs and symptoms of uterine fibroids | Sep 2018 | 334 days |
*Negotiation initiation and completion/closed date assumed to be mid-month; approximate calendar days calculated.
Closed
| Brand Name | Generic Name | Manufacturer | Indication | Initiation | Duration* |
|---|---|---|---|---|---|
| Monoprost | Latanoprost | Laboratoires Théa | Glaucoma and ocular hypertension | Sep 2018 | 334 days |
Signals Decoded:
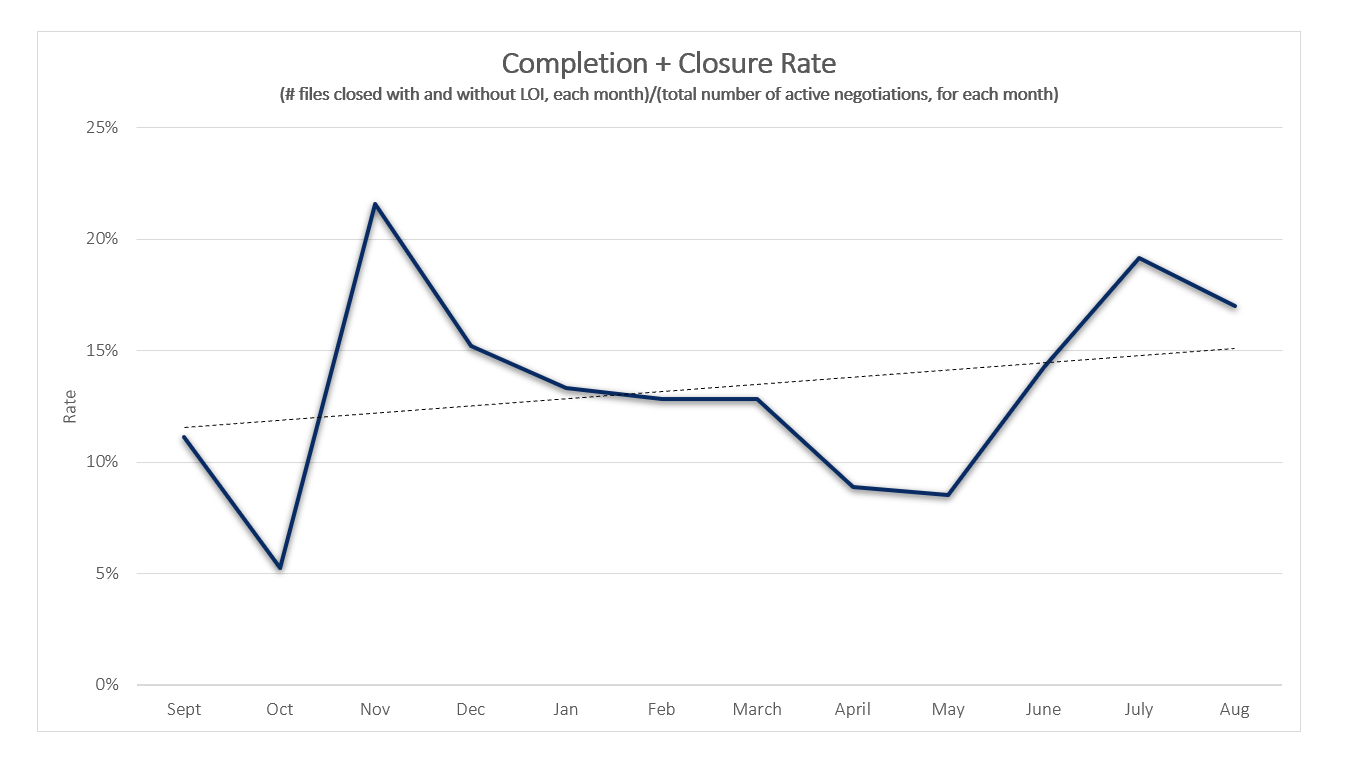
August saw the completion of the second bevacizumab biosimilar, along with 4 other oncology files – all reaching agreement within 2-3 months of negotiations. Two files that had been under negotiations for almost a year were also finalized, resulting in a completed agreement for Fibristal, and closed negotiations for Monoprost.
Negotiation Initiation
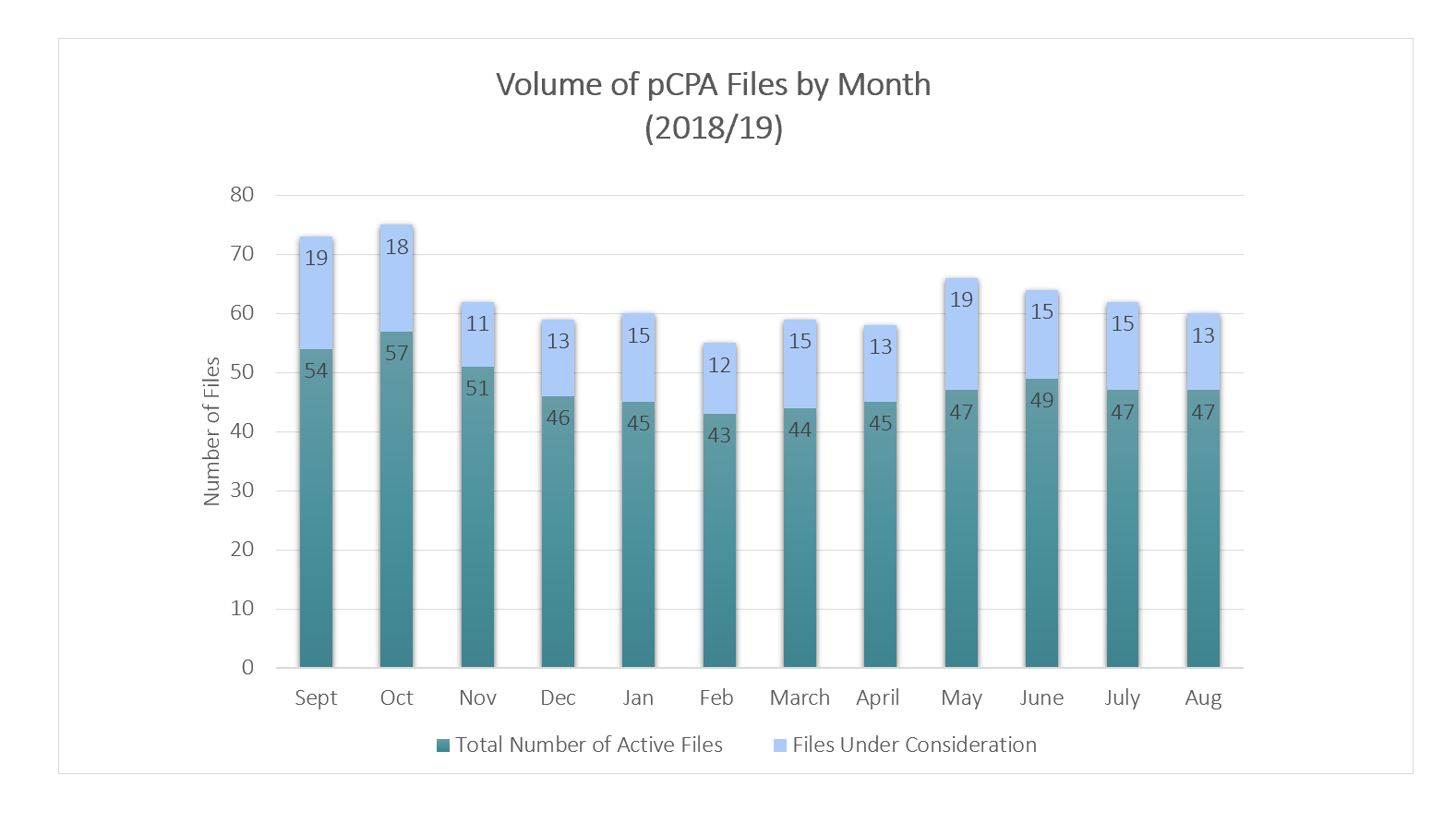
The pCPA initiated 8 new negotiations since the last update, for a total of 47 active negotiations.
| Brand Name | Generic Name | Manufacturer | Indication | CADTH Date | Initiation Time* |
|---|---|---|---|---|---|
| Bosulif | Bosutinib | Pfizer | Chronic Myeloid Leukemia | 2 Aug 19 | 13 days |
| Emerade | Epinephrine bitartrate | Bausch Health | Emergency treatment of anaphylactic reactions | N/A | N/A |
| Ibrance/Faslodex | palbociclib/fulvestrant | Pfizer | Advanced or Metastatic Breast Cancer | 21 May 19 | 86 days |
| Keytruda | Pembrolizumab | Merck | Metastatic non-squamous non-small cell lung cancer | 17 Jun 19 | 59 days |
| Orfadin Oral Suspension | Nitisinone | SOBI | Hereditary tyrosinemia type 1 (HT-1) | N/A | N/A |
| Vizimpro | Dacomitinib | Pfizer | Locally advanced/metastatic non‑small cell lung cancer with epidermal growth factor receptor-activating mutations | 17 Jun 19 | 59 days |
| Vyzulta | Latanoprostene bunod | Bausch Health | Open-angle glaucoma or ocular hypertension | 24 Jul 19 | 22 days |
| Xalkori | Crizotinib | Pfizer | ROS1-positive advanced Non-Small Cell Lung Cancer | 7 Jun 19 | 69 days |
*Negotiation initiation and completion/closed date assumed to be mid-month; approximate calendar days calculated.
Signals Decoded:
Keytruda is now being negotiated for 3 different indications and is under consideration for a fourth. Negotiations for Bosulif were initiated the same month as the recommendation was issued – a review triggered by a Drug Program Request for Advice. Finally, Ibrance, Xalkori and Orfadin have all completed previous pCPA negotiations.
Files Under pCPA Consideration
6 new drug products, including one initiated by a Drug Program Request for Advice, received a CDEC recommendation or pERC notification to implement in August 2019, for a total of approximately 13 products under pCPA Consideration.
| Brand Name | Generic Name | Manufacturer | Indication | Recommendation |
|---|---|---|---|---|
| Alunbrig | Brigatinib | Takeda | Non-Small Cell Lung Cancer (NSCLC) |
Do not reimburse
|
| Lenvima | lenvatinib | Eisai | Hepatocellular Carcinoma | Conditional |
| Lutathera | Lutetium Lu 177 dotatate | Advanced Accelerator Applications | Gastroenteropancreatic neuroendocrine tumors | Conditional |
| Olumiant | baricitinib | Eli Lilly | Rheumatoid Arthritis | Conditional |
| Bosulif* | bosutinib | Pfizer | Chronic Myeloid Leukemia | Recommend |
| Keytruda | Pembrolizumab | Merck | Melanoma Adjuvant Treatment | Conditional |
| Onpattro** | patisiran | Alnylam Netherlands BV | Polyneuropathy in hereditary transthyretin-mediated amyloidosis | Conditional |
*Request for Advice
**Correction: Onpattro received a final recommendation in July and was not included in the MORSE July pCPA Update
Not Negotiated
| Brand Name | Generic Name | Manufacturer | Indication | CADTH Date | Time to Decision* |
|---|---|---|---|---|---|
| Ninlaro | Ixazomib | Takeda | Multiple Myeloma (2nd-beyond) | July 22, 2019 | 24 days |
*Negotiation initiation and completion/closed date assumed to be mid-month; approximate calendar days calculated.
To receive updates similar to this one please be sure to sign up for MORSE Signals, our free e-mail newsletter service.







